 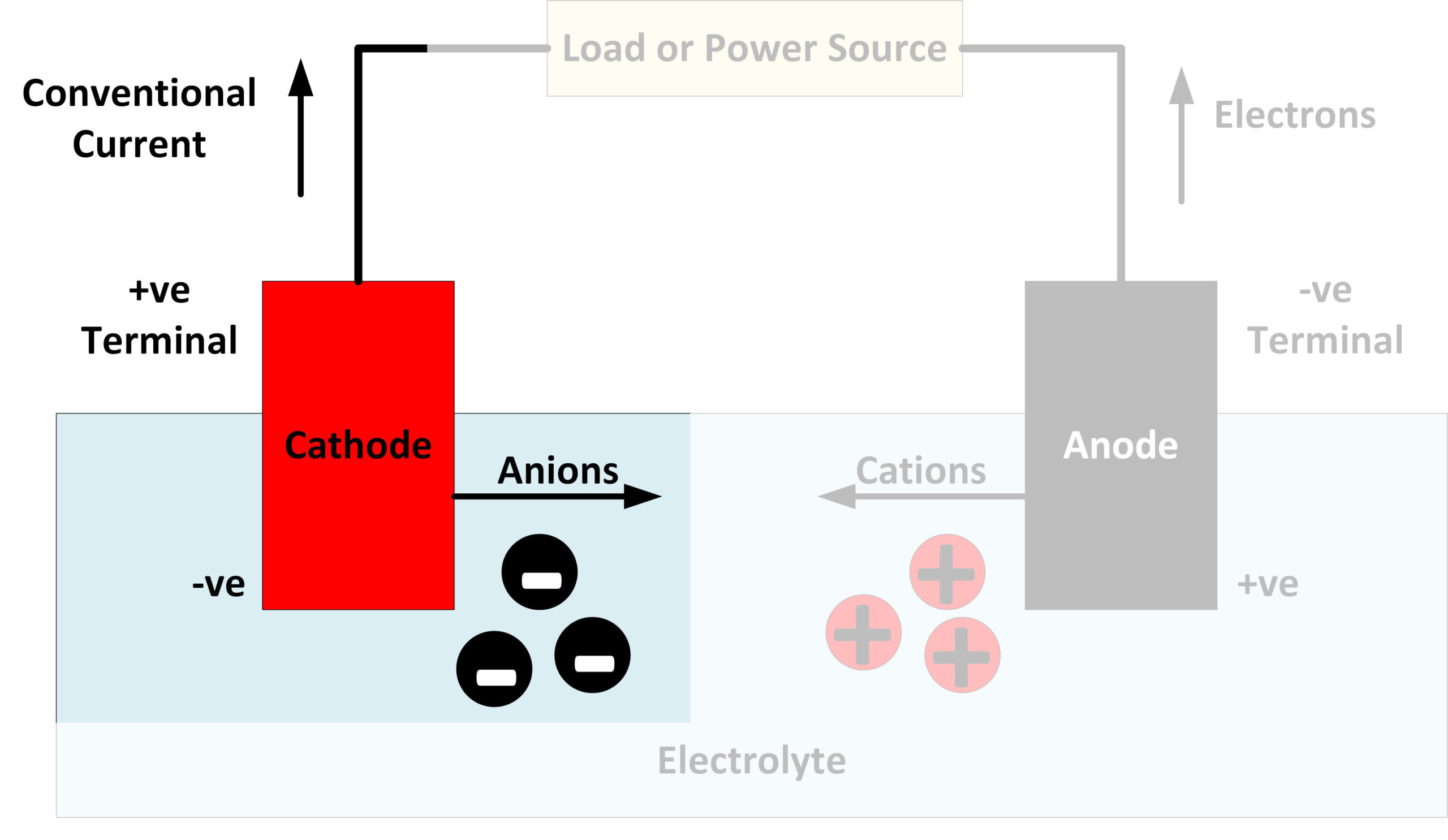
As the battery is discharged, the process is reversed, as shown in the following formula. In the anode, cadmium hydroxide is transformed to cadmium. The most commonly used battery of all is an alkaline. The half-reactions that occur in ordinary alkaline batteries can be written asĬathode: $\mathrm)$. When the battery is charged, the chemical composition of the cathode is transformed and the nickel hydroxide changes to nickel oxyhydroxide NiOOH. Eventually they can no longer retain a charge and are considered dead. Liquid alkaline electrolyte is then injected into the case and given time to be absorbed by the separator. An ion conducting sheet of paper is used as the separator. The case is then indented to hold the cathode in place. Number of coulombs of charge released by the reduction of 10.0 g of MnO2 to MnO(OH). The cathode is a manganese based hollow cylinder which is inserted into the case. Quantitatively compare the number of coulombs of charge released by the reduction of 10.0 g of K2FeO4 to Fe(OH)3 with the Each cell consists of an anode, a cathode and an electrolyte material that facilitates electron. In alkaline batteries, the cathode is made of manganese dioxide and the anode is made of zinc. (c) A "super-iron" battery should last longer than an ordinary alkaline battery of the same size and weight because its cathode can provide more charge per unit mass. Alkaline batteries contain essentially a chemical reaction. How Do Alkaline Batteries Work Batteries contain two terminals: a positively charged cathode and a negatively charged anode, with a chemical electrolyte positioned between them that allows ions and electrons to flow. The halfreaction occurs in a basic environment. (b) Write a balanced equation for the cathode halfreaction in a "super-iron" battery. (a) Use the following standard reduction potential and any data from Appendixes C and D to calculate the standard cell potential expected for an ordinary alkaline battery: Moreover, the experimental results indicate that charging time for an alkaline battery using doped Mn 3 O 4 cathode could possibly shorten to as little as 30 min. This battery uses the same anode reaction as an ordinary alkaline battery but involves the reduction of FeO4^2- ion (from K2FeO4 ) to solid Fe(OH)3 at the cathode. The doped cathode can also maintain a cycling capacity of 261 mAh g 1 while holding a 95.3 reversible capacity after 60 cycles at a high rate of 500 mA g 1. In 1999, researchers in Israel reported a new type of alkaline battery, called a "super-iron" battery. SOLVED: The half-reactions that occur in ordinary alkaline batteries can be written as 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
